Finely dispersed instead of clumped together
Neumann Group develops New Metal Phosphide Catalyst
The research group led by Lise Meitner group leader Dr. Constanze Neumann has developed a new type of catalyst that could be of interest to the chemical industry. The scientists have published their findings in the journal "Angewandte Chemie International".

Transition metal phosphides have been dreamed of as a potential replacement for noble metal catalysts for a while now. They are cheaper and available in sufficient quantities. “But there have always been challenges with the surface oxidation and synthesis”, says Dr. Constanze Neumann, Lise-Meitner group leader at the Max-Planck-Institut für Kohlenforschung in Mülheim. For example, their production required very high temperatures or reactive starting material that might be costly, highly toxic – or both. Together with her team, the young chemist has found an elegant method to provide a single-step procedure that relies on both safe and cheap materials to synthesize an air-stable, nickel-containing catalyst. The group has published their results as a “Very important Paper” with Angewandte Chemie International.
“In our paper we show that it is possible to reach a high dispersion of the catalyst”, says Dr. Leila Karam, Postdoc in Constanze Neumann’s group and first author of the paper. With the help of the right surface ligand, the scientists are able to apply their catalyst to a carrier surface with the broadest possible distribution - and in such a way that it is available for the desired reaction without clumping or oxidizing beforehand. The ligands are able to protect the catalyst from undesired oxidation but do not hinder it in promoting the desired reaction. This fine distribution ensures that the catalyst can be used in just as small quantities as conventional alternatives containing palladium.
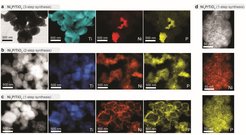
“We are very pleased that our catalyst can keep up with commercial palladium catalysts”, says Constanze Neumann. This might be interesting for chemical companies, where heterogeneous palladium catalysts are widely used for selective hydrogenation, including in the synthesis of pharmaceuticals.
And there is another special feature: "We were able to show that our catalyst still works well even if it was stored under air for more than a year. The material is not rendered unusable by oxidation," says Constanze Neumann. This makes both the storage and handling of the catalyst much easier: while other phosphides often have to be worked with in the glovebox, the Mülheim catalyst can be handled in a normal fume hood.
Happy as they are with their findings, the group does not want to stop there: “We want to improve the reusability of our catalyst”, states Constanze Neumann. Furthermore, the researchers would like to get rid of any solvents that are needed to produce their catalyst. “We would like to keep the production as green as possible!”




